|
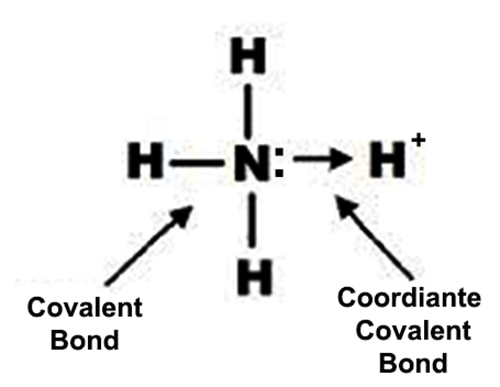
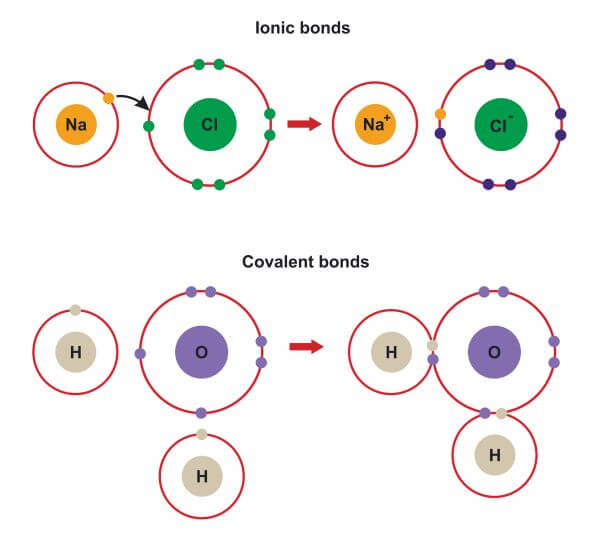
Furthermore, many covalent compounds are volatile, whereas ionic compounds are not.ĭespite the differences in the distribution of electrons between these two idealized types of bonding, all models of chemical bonding have three features in common: (For more information about solution conductivity, see Chapter 4 "Reactions in Aqueous Solution", Section 4.1 "Aqueous Solutions".) In contrast, most covalent compounds that dissolve in water form solutions that do not conduct electricity. Ionic compounds, for example, typically dissolve in water to form aqueous solutions that conduct electricity. The concepts of covalent and ionic bonding were developed to explain the properties of different kinds of chemical substances. We also introduced two idealized types of bonding: covalent bonding A type of chemical bonding in which electrons are shared between atoms in a molecule or polyatomic ion., in which electrons are shared between atoms in a molecule or polyatomic ion, and ionic bonding A type of chemical bonding in which positively and negatively charged ions are held together by electrostatic forces., in which positively and negatively charged ions are held together by electrostatic forces. In Chapter 2 "Molecules, Ions, and Chemical Formulas", we defined a chemical bond as the force that holds atoms together in a chemical compound. To present three common features of chemical bonding.As you learn about bonding, you will also discover why, although carbon and silicon both have ns 2 np 2 valence electron configurations and form dioxides, CO 2 is normally a gas that condenses into the volatile molecular solid known as dry ice, whereas SiO 2 is a nonvolatile solid with a network structure that can take several forms, including beach sand and quartz crystals. Moreover, you saw in Chapter 7 "The Periodic Table and Periodic Trends" that diamond is a hard, transparent solid that is a gemstone graphite is a soft, black solid that is a lubricant and fullerenes are molecular species with carbon cage structures-yet all of these are composed of carbon. For example, oxygen gas (O 2) is essential for life, yet ozone (O 3) is toxic to cells, although as you learned in Chapter 3 "Chemical Reactions", ozone in the upper atmosphere shields us from harmful ultraviolet light. What you learn in this chapter about chemical bonding and molecular structure will help you understand how different substances with the same atoms can have vastly different physical and chemical properties. Silicon dioxide is a giant covalent structure, whose strong bonds in three dimensions make it a hard, high-melting-point solid, such as quartz. Because of its simple molecular bond, carbon dioxide is a gas that exists as a volatile molecular solid, known as “dry ice,” at temperatures of −78☌ and below. Both the group 14 elements carbon and silicon form bonds with oxygen, but how they form those bonds results in a vast difference in physical properties. The properties of these substances depend on not only the characteristics of the component atoms but also how those atoms are bonded to one another.Ĭarbon and silicon bonding. The properties described in Chapter 6 "The Structure of Atoms" and Chapter 7 "The Periodic Table and Periodic Trends" were properties of isolated atoms, yet most of the substances in our world consist of atoms held together in molecules, ionic compounds, or metallic solids. Our goal is to understand how the properties of the component atoms in a chemical compound determine the structure and reactivity of the compound. In this chapter and Chapter 9 "Molecular Geometry and Covalent Bonding Models", we describe the interactions that hold atoms together in chemical substances, and we examine the factors that determine how the atoms of a substance are arranged in space. 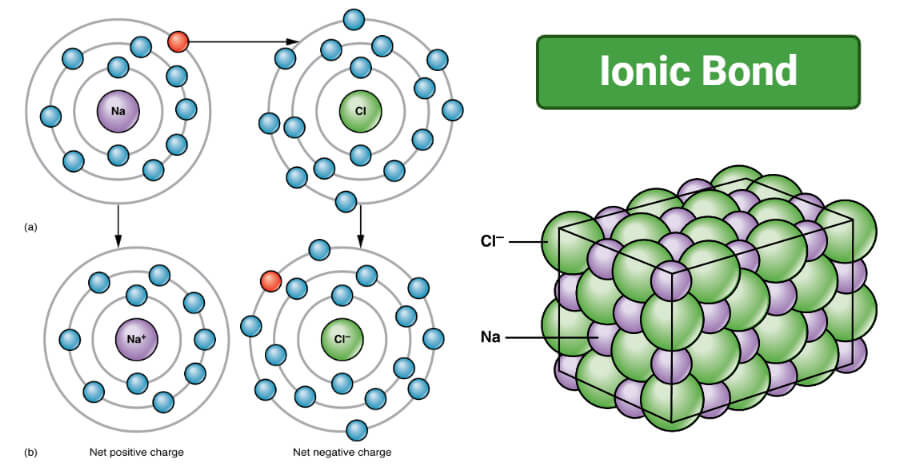
It is caused by the lasting attraction between positive and negative charges.In Chapter 7 "The Periodic Table and Periodic Trends", we described the relationship between the chemical properties and reactivity of an element and its position in the periodic table. Saturated Unsaturated and Supersaturatedīonding is the interaction of different atoms to form compounds, molecules, ions, crystals, and all the other substances that make up the world.Reaction Quotient and Le Chatelier's Principle.Prediction of Element Properties Based on Periodic Trends. 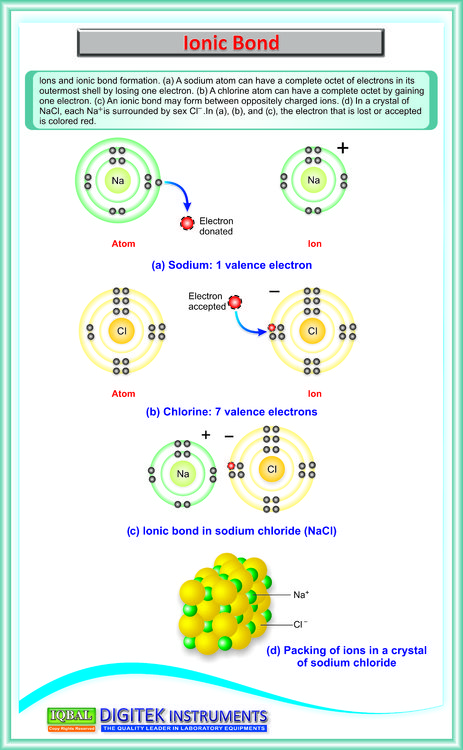
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
